
What is the molar mass of Mg3 PO4 2?īy balancing the charges, the chemical formula of magnesium phosphate would be Mg3(PO4)2. We need to find out how many moles are in 55.7 g of HGO. Is 216.589 grams, That is, one mole of HGO = 216.589 grams of HGO. The periodic table also tells us that the atomic mass of an O is 15.999. This red or yellow crystalline solid is also used as an electrode (mixed with graphite) in zinc-mercuric oxide electric cells and mercury batteries. Mercury(II) oxideHGO provides elemental mercury for the preparation of various organic mercury compounds and some inorganic mercury salts. The mass of 2 moles of HgO is 433.18g, What is HgO in Chemistry? So, two moles would be twice the molar mass of 1 mole of HgO. You can get that by adding the molar mass of Hg to the molar mass of O. View your results with Omni Calculator.Divide the weight by the atomic or molecular mass.Find its atomic or molecular mass using the periodic table.What is the quantity required in mL of 0.716 M KBr solution to provide 30.5 g of KBr?Īnswer: The answer is: the volume of kbr is 357 ml, How do I Calculate Moles? What is the formula for grams to moles? thread related links Then the mass of the gas divided by the moles will give the molar mass. The ideal gas law will be used first to solve for the mole of the unknown gas (n). See also Does Loremaster Chest Drop Spells? How do you find the molar mass of a gas? M is the molar mass of the substance (mass of one mole of the substance) in g mol–1. m is the mass of the substance, in grams, g. N = m/m n is amount of substance, in moles, mol. Now, the molar mass of potassium bromide is 119.002 g mol−1 Which means that each mole of potassium bromide has a mass of 119.002 g.
What is the mass in grams of KBr in a solution? However, the molar mass can also be calculated as Multiplying the atomic mass in amu by the molar mass constant (1 g/mol), To calculate the molar mass of a compound containing many atoms, sum all the atomic masses of the constituent atoms.
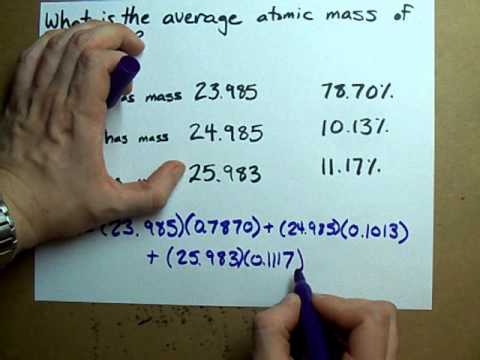
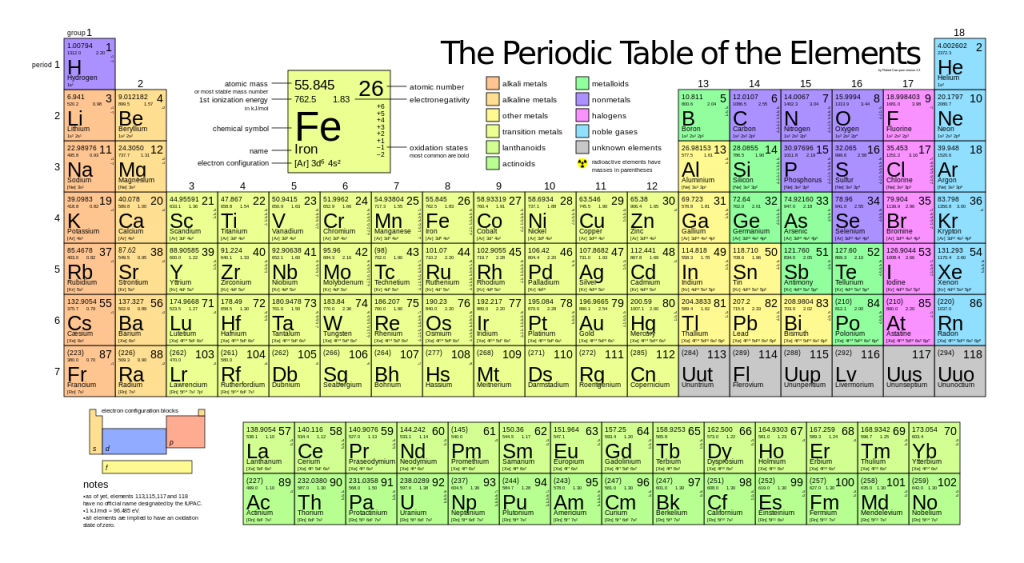
The characteristic molar mass of an element is simply the atomic mass in g/mol.
